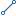| Description | The textual representation of the planned activity from the point of view of a blinded participant (study subject or study investigator).
For example:
During the second treatment epoch of a study, Arms A and B are still blinded and Arm C is no longer blinded. So, Arm A and B must have identical blinded descriptions.
In a study with 3 arms, Arm 1: standard vaccine given in three shots at 2 months, 5 months, and 12 months of age; Arm 2: new vaccine given in three shots at 2 months, 5 months, and 12 months of age; Arm 3: new vaccine given in two shots at 2 months and 5 months of age. Subjects assigned to the third arm are unblinded at some point during 5 months and 12 months. By the time of the Third Shot Epoch, the "Arm 3/Third Shot" activity can be called by this, its unblinded name. However, the "Arm 1/Third Shot" and "Arm2/Third Shot" activities still need blinded names. Both these activities would have the blinded name (something like) "3-shot Arm/Third Shot". |
 Activity Nm
Activity Nm Activity Sk
Activity Sk Agent Administration Care Setting Type Code Sk
Agent Administration Care Setting Type Code Sk Blinded Descr
Blinded Descr Category Code Sk
Category Code Sk Comment Txt
Comment Txt Description Txt
Description Txt Duration Qty
Duration Qty Effective From Dt
Effective From Dt Effective To Dt
Effective To Dt Identification Num
Identification Num Interruptible Ind
Interruptible Ind Load Info Sk
Load Info Sk Purpose Txt
Purpose Txt Reason Code Sk
Reason Code Sk Repeat Duration Qty
Repeat Duration Qty Repeat Frequency Code Sk
Repeat Frequency Code Sk Repeat Frequency Ratio
Repeat Frequency Ratio Repeat Quantity Range
Repeat Quantity Range Source Code Sk
Source Code Sk Status Change Reason Code Sk
Status Change Reason Code Sk Status Code Sk
Status Code Sk Status Dt
Status Dt Study Accrual Threshold Pct
Study Accrual Threshold Pct Study Day Range Qty
Study Day Range Qty Study Focus Ind
Study Focus Ind Study Site Accrual Threshold Pct
Study Site Accrual Threshold Pct Subcategory Code Sk
Subcategory Code Sk Tenant Sk
Tenant Sk Valid From Ts
Valid From Ts Valid To Ts
Valid To Ts